Installation, in five steps.
From the IFU, Section 4. The device must be correctly seated and stable within the drain system before completing installation.
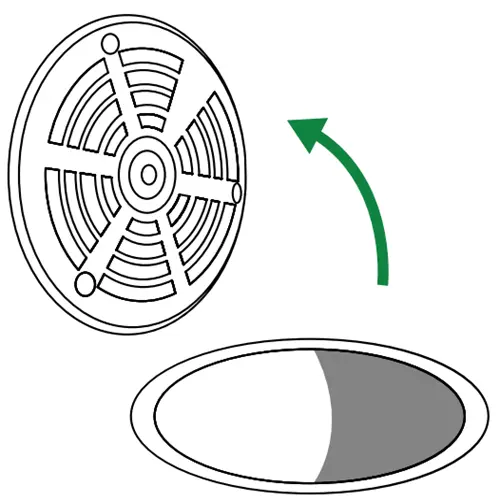
Remove existing drain grate
Lift out the grate to expose the drain opening. Set the grate and any screws aside in a safe place; you will reinstall these once the device is in position.
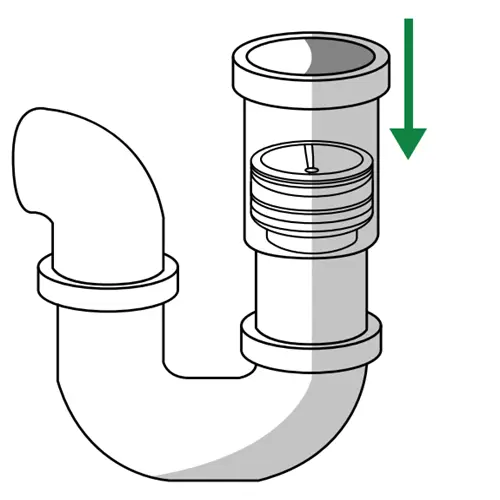
Insert Green Drain into the drain opening
Position the device with the silicone skirt downward and push it straight into the drain opening. The flexible housing compresses slightly for a friction fit inside the pipe; no adhesive or sealant is required.

Position within the pipe as appropriate
The IFU diagram indicates a recess of 2" to 4" into the physical pipe, depending on drain configuration. Recessing keeps the device clear of the grate above and lets the original grate sit flush.
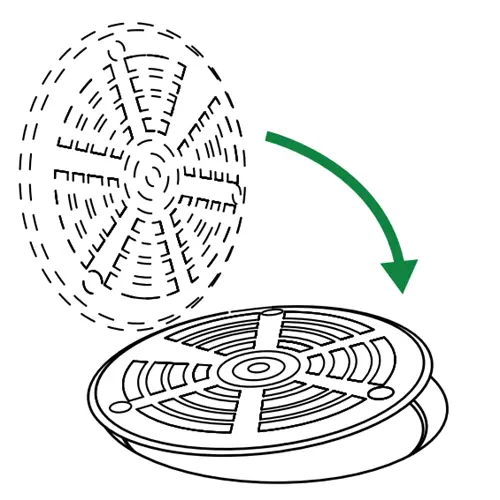
Replace and close the drain grate
Reseat the original grate over the device. Because the Green Drain sits inside the pipe, the grate fits exactly as it did before, with no visible change to the drain.
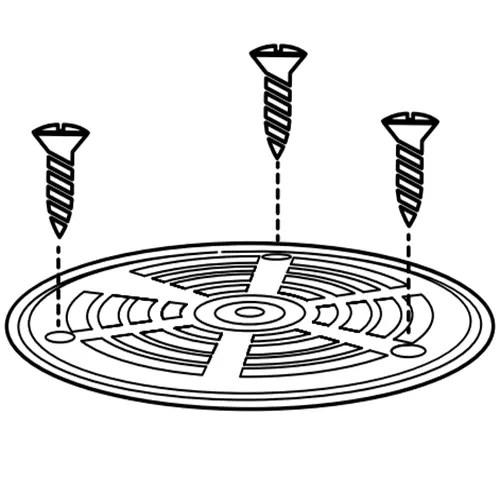
Secure using screws if applicable
Some grates require screws to lock in place. Many floor drains use gravity-set grates that do not require screws. Either way, confirm the device remains correctly positioned after fastening.
A complementary general installation guide is available on the Installation Guide page.